
Marking of charges are significantīecause it is used to determine the best lewis structure of the ion. Now there is no more lone pairs to mark on carbon atomsĪfter marking on oxygen atoms (outside atoms).Īfter, marking electron pairs on atoms, we should mark charges of each atom. For three oxygen atoms, nine electrons pairs are spent.Will take three lone pairs following the octal rule (oxygen atom cannot keep more than First, mark those nine valence electrons pairs as lone pairs in outside atoms (on oxygen atoms).Therefore only nine (12-3 = 9) valence electrons There are already three C-O bonds in the above sketch.Carbon has the moreĬhance to be the center atom (See the figure) because carbon can show valance of 4. To be the center atom, ability of having higher valance is important. For,ĬO 3 2- ion, Total pairs of electrons are 12. Total electron pairs are determined by dividing the number total valence electrons by two. Total valance electrons pairs = σ bonds + π bonds + lone pairs at valence shells Total valence electrons = 4 + 18 + 2 = 24.Therefore there are two more electrons which Total valence electrons given by oxygen atoms = 6 *3 = 18.There are three oxygen atoms in CO 3 2- ion, Therefore Total valence electrons given by carbon atom = 4.So, carbon has four electrons in its valence shell.Oxygen Total number of electrons of the valance shells of CO 3 2-Ĭarbon is located at group 4 in the periodic table. Check the stability and minimize charges on atoms by converting lone pairs to bonds.ĭrawing correct lewis structure is important to.Find total number of electrons of the valance shells of carbon and oxygen atoms.The orbitals would not be able to overlap, so the connection between the atoms would be lost.Steps of drawing lewis structure of CO 3 2-įollowing steps are required to draw the CO 3 2- lewis structure and they are explained in detail If one atom turns with respect to the other, the p orbital would have to stretch to maintain the connection. A s orbital is not affected when the atom at one end of the bond is rotated with respect to the other. In other words, there are more nodes in the higher-energy orbitals than in the lower-energy ones.Īn important consequence of the spatial distribution or "shape" of a p orbital is that it is not symmetric with respect to the bond axis. Just as the sigma-bonding orbitals display progressively shorter wavelengths along the bonding axis as they go to higher energy, so do the pi bonding orbitals. There will be both bonding and antibonding combinations. The two p orbitals orthogonal to the bond axis can engage in p bonding. In a main group diatomic species like dinitrogen, one p orbital lying along the bond axis can engage in s bonding. A similar picture could be shown for the other set of p orbitals. The second picture shows the result of the constructive (or destructive) interference. The illustration above is for one set of p orbitals that are orthogonal to the bond axis. the electron density is found above and below the bond axis.the resulting orbitals contain nodes along the bond axis.parallel p orbitals can overlap to produce bonding and antibonding combinations.  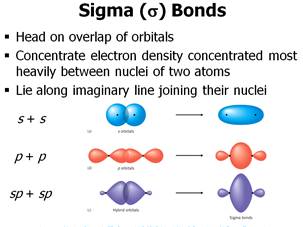 
They could also make an out-of-phase combination, as shown below. They can make an in-phase combination, as shown below. They can be close enough to each other to overlap, although they do not overlap as strongly as orbitals lying along the bond axis. They would approach each other side by side, above and below the bond axis between the two atoms. Parallel, but not collinear, p orbitals can also interact with each other. \)Įarlier, we saw that p orbitals that lie along the same axis can interact to form bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
